Hello flow cytometry fans, Wolfie here with more tips to help you improve your cell sorting experiments. To paraphrase Captain Salazar “dead cells tell the wrong tales.”[1] That is to say, dead cells can take up all the antibodies in the staining mix and mimic a live cell. The consequences of this can be complex and costly, depending on what the cells are being used for after sorting.
When cells die, one of the processes that happens is that the membrane becomes compromised, allowing transport across the previously impermeant barrier. This is why antibodies in the staining buffer can get captured inside the cell, mimicking a live cell. Think about the possibility that there are 10% dead cells, but the population of interest is at 1% or lower. The numbers alone support the need to get rid of dead cells.
One of the most common ways to do this is to use a cell impermeant dye. These compounds cannot get into cells with an intact membrane. The most common class of these dyes are the nuclear dyes, as listed in table 1.
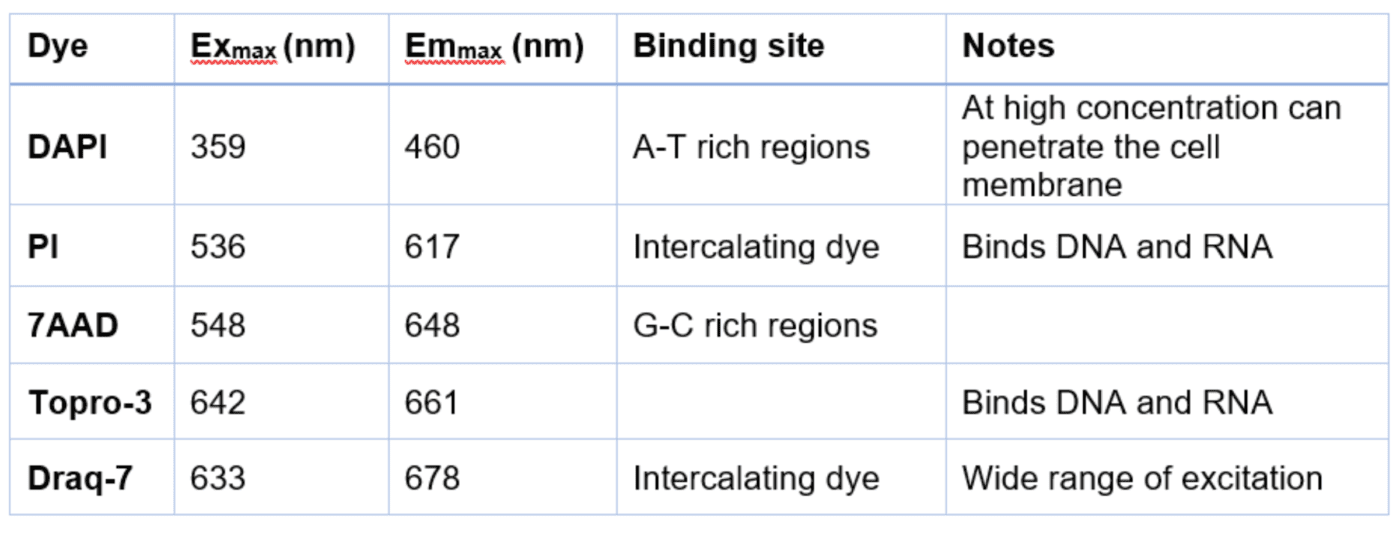
Table 1: Commonly used dyes to access cell viability in flow cytometry
*Note this list is not exhaustive, but a collection of the most commonly used dyes
In each case, these dyes are not fluorescent by themselves and are generally weakly fluorescent when bound to DNA. However, the amount of DNA in the cells makes the signal from these compounds very intense. As table 1 shows, these come in a range of excitation and emission profiles. Typical data is shown in figure 1.
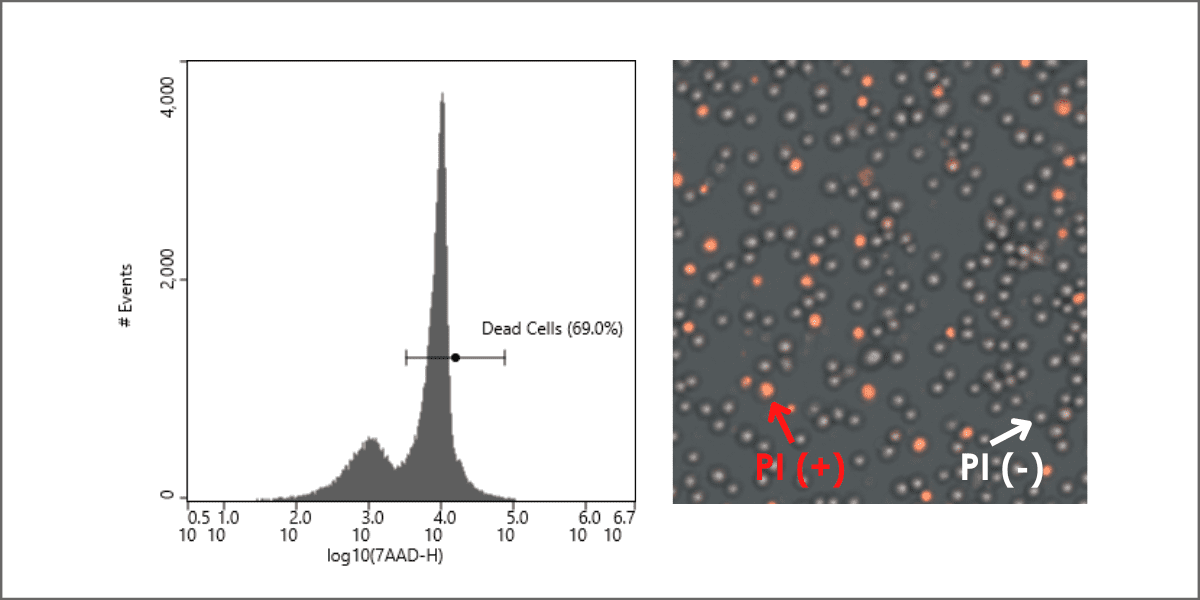
Figure 1: Typical representation of cell viability on an histogram
plot where dead cells are positively stained with 7AAD dye.
As you can see from this figure, the positive cells are eliminated as dead cells, and the rest are carried forward for further gating to identify the target cells.
Another option for identifying live and dead cells are the amine-reactive dyes. These dyes bind to amine groups on proteins. On an intact cell, there are few amine groups on the surface, so there is low-level of binding. With dead cells, these dyes penetrate the membrane and inside the cell is a lot of amine groups, so there is a very bright signal. These dyes are very useful for intracellular staining protocols, but are less commonly used for cell sorting. One thing to remember with these dyes is that they must be stained in the absence of protein. That BSA in your staining buffer will bind all the amine reactive dye, leaving your dead cells unstained. You can check out these amine reactive dyes (known as the Zombie dyes) from our partner BioLegend.
A third option to consider is to stain live cells, rather than dead cells. This can be accomplished using a dye such as Calcein AM (Exmax 493/Emmax 519). This is a cell permeant dye and when it enters the cell, it is cleaved by cellular esterases, making the dye fluorescent. Since dead cells leak esterase, they are not fluorescent.
While most cell impermeant nuclear dyes will work for downstream sequencing applications, Illumina suggests for their multiome assay one should use 7-AAD as a viability dye. This article also points out that DAPI at high enough concentration can enter live cells, which could negatively impact your sequencing run. They also caution against cell permeant dyes, however, as those bind to DNA and can interfere with downstream sorting applications.
Removing dead cells is a critical step for having good analysis, and you now have several different tools to identify these cells in your experiment. With the wide range of excitation and emission profiles, there clearly is one to fit your needs.
Thanks for reading! Until next time, keep flowin’.
–Wolfie
[1] The actual quote, from ‘Pirates of the Caribbean: Dead Men Tell No Tales’ is “I wish I could tell him myself, but dead men tell no tales.”





