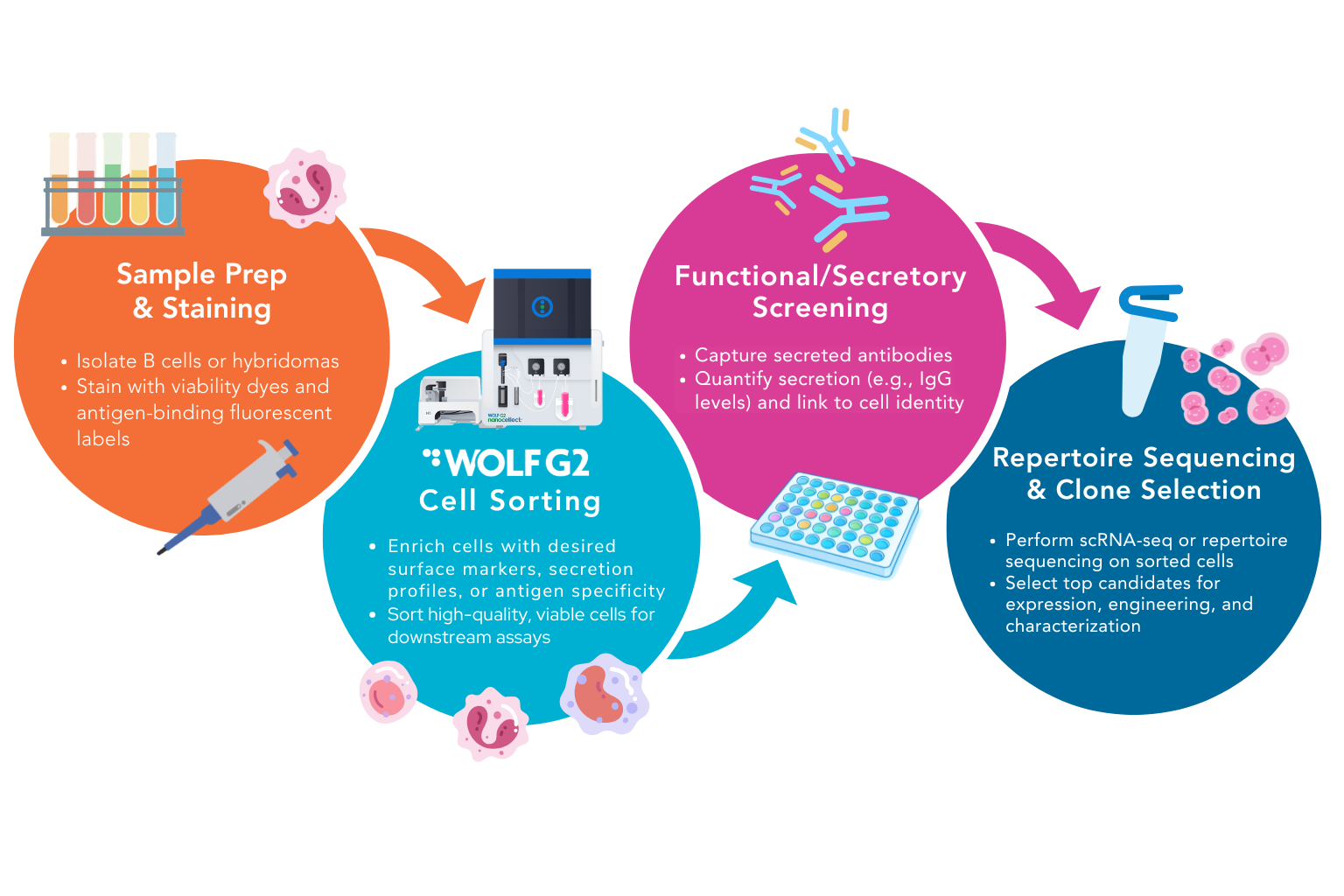
Antibody discovery is a cornerstone of modern biotechnology, underpinning everything from therapeutic mAb (monoclonal antibody) drugs to diagnostic reagents and immune profiling tools. At its core, antibody discovery depends on isolating, characterizing, and optimizing cells (often B cells or hybridomas) that produce the desired antibodies with high affinity and specificity. To power high-resolution downstream analyses—especially single-cell sequencing, functional screening, and secretory phenotyping—researchers must start with highly purified, viable cells. This is where the WOLF G2 antibody discovery workflow offers a significant advantage for scientists involved in these processes.
The WOLF G2® Cell Sorter uses gentle microfluidic sorting technology that preserves cell viability and phenotype while delivering enrichment of target populations. Its low-pressure, disposable cartridge system minimizes cell stress compared with traditional high-pressure droplet sorters, making it optimal for sensitive primary cells, immune cells, and B cells used for WOLF G2 antibody discovery workflows.
Key advantages include:
Low shear stress sorting suitable for fragile antibody-producing cells.
High sensitivity detection and sorting with multi-fluorescent panels.
Compatibility with single-cell genomics and proteomic workflows.
Single-cell deposition into plates for clonal expansion or functional assays.
Li et al., New Biotechnology 2025 demonstrates the use of WOLF sorting during early antibody discovery steps. In this study the authors developed site-directed mAbs targeting the KRAS G12D driver mutation, using flow cytometry to enrich live, marker-defined B cell populations before characterization of binding specificity. The WOLF sorter helped isolate phenotypically defined populations critical for validating their antibody candidates prior to downstream biochemical assays. – Read the Article
Why it matters: Robust sorting of live cells with correct antigen specificity is essential for generating high-quality monoclonal antibodies against challenging targets like KRAS.
Cheng et al., Nature Communications (2023) introduced SEC-seq, a method that directly associates the amount of secreted IgG (or other protein) with single-cell transcriptomes and surface markers in human plasma cells. This approach uses nanovials that capture secretions and then integrates flow cytometry and single-cell RNA sequencing to connect function (antibody secretion) with gene expression profiles. Importantly, the workflow uses WOLF cell sorter to enrich viable secretory cells prior to library preparation and sequencing. – Read the Article
Why it matters: Techniques like SEC-seq depend on precise enrichment of secretory cells so that high-quality transcriptomic profiles can be linked with secretory output—a powerful paradigm for selecting cells producing antibodies with desired functional properties.
The Cell article Rosen et al., Cell 2024 reports on the isolation of broadly neutralizing antibodies against SARS-related viruses, leveraging high-throughput profiling strategies. While the paper itself focuses on antibody characterization and neutralization data, much of the underlying discovery pipeline—including sorting of immune cells with desired specificity—relies on precise phenotypic selection by flow cytometry as an upstream step to functional screening and sequencing. – Read the Article
Why it matters: High-throughput functional screens in emerging infectious diseases depend on isolating B cells with rare neutralizing responses—a task where gentle, flexible sorting platforms like WOLF are invaluable.
Antibody discovery today often moves beyond simple bulk binding assays—especially when the goal is deep functional profiling of immune repertoires. Integration of phenotypic sorting with modern single-cell multi-omics (e.g., transcriptome + surface proteins + secretory output) allows researchers to:
Identify rare antibody-producing cells from complex immune populations.
Correlate genetic signatures with functional secretion (e.g., high IgG producers).
Discover new surface markers predictive of desired functional phenotypes.
Seed single-cell libraries for paired heavy + light chain sequencing.
The landscape of antibody discovery has evolved rapidly—from hybridomas to high-throughput single-cell techniques linking phenotype, genotype, and function in unprecedented detail. Across different phases of this pipeline, the WOLF G2 antibody discovery workflow offers a gentle, reliable means to enrich target cell populations while preserving cellular integrity for downstream molecular analysis. Its compatibility with modern workflows like SEC-seq and its effectiveness in isolating rare antigen-specific cells make it a valuable component of next-generation antibody discovery platforms.





